posted 11-04-98 06:04 AM
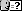
2-acetamidohexane [N-(2-hexyl)acetamid]
To a ice-cooled solution of 64.8g (0.20mol) of Hg(NO3)2 in 100ml MeCN
16.8g (0.20mol) of 1-hexene are slowly added keeping the temp below 30°C.
The clear yellow solution is stirred for 1hr at RT. With ice-cooling 200ml
3N NaOH are added, followed by a solution of 3.80g (0.10mol) of NaBH4 in
200ml 3N NaOH. Again stir for another hour.
Decant the aq/organic layer
leaving the elemental Hg behind, saturate the aq. layer with NaCl and
extract with Et2O (2*100ml). The extracts are dried with Na2SO4, the
solvent removed and the residue distilled. 22.8g (80%) amide, bp (1.6
torr) 93-96°C.
Ref: JACS 91, 5647 (1969)
Hydrolysis (use the crude amide):
20.0g (0.14mol) of the above
amide, 15.7g (0.28mol) KOH in 25ml H2O and 62.5 ml ethyleneglycol are
refluxed for 24 hours with good stirring.
After cooling to RT extract
with ether (3*100ml), back-extract the amine with 3*100ml 2N HCl. From the
ether 3.08g of unconverted amide are obtained.
The acidic aq. extracts
are basified with 30g NaOH and the amine is extracted with ether
(2*150ml). The extract is dried with K2CO3, the ether evaporated and the
crude amine is distilled. 7.50g (53%, 62% considering the unreacted
amide), bp 114-116°C.

 the
Hive BB
the
Hive BB 
 Chemistry
Discourse
Chemistry
Discourse 
 Solvomercuration-demercuration
Solvomercuration-demercuration